
In these molecules, the polarizability is greatest when the field is applied parallel to the molecule rather than perpendicular. A modern periodic table arranges the elements in increasing order of their atomic numbers and groups atoms with similar properties in the same vertical column (Figure 3.2b). Molecular Orientationįor unsaturated molecules, like 2,4 hexadiene, the molecular orientation is significant. In chemistry, a family is a group of elements with similar chemical properties.Chemical families tend to be associated with the vertical columns on the periodic table.The term 'family' is synonymous with the term 'group'. The trend across a period is complicated and explained in our article on ionic radius. Hence, the polarizability also increases down a group. Electrons in paired spin configurations are slightly easier to. Electrons in an s s orbital can shield p p electrons at the same energy level because of the spherical shape of the s s orbital. We know that the ionic radius increases down a group. Electron shielding refers to the blocking of valence shell electron attraction by the nucleus, due to the presence of inner-shell electrons. Since large atoms are easily polarizable, the polarizability trend in the periodic table is similar to that of ionic radius. Most periodic tables provide additional data (such as atomic mass) in a box that contains each element’s symbol. A modern version is shown in Figure 4.6.2 4.6. These atoms and ions can be easily polarized. The periodic table is one of the cornerstones of chemistry because it organizes all of the known elements on the basis of their chemical properties. On the other hand, large atoms and anions with more electrons have large radii and diffused electron clouds that weaken the nucleus-electron interactions. An element group is a vertical column on the. Element atomic number increases as you move down a group from top to bottom or across a period from left to right. Both groups and periods reflect the organization of electrons in atoms. The periodic table organizes elements into vertical groups. These smaller atoms are generally challenging to polarize. A group is a vertical column down the periodic table, while a period is a horizontal row across the table. The periodic table is the chemists best tool for figuring out charges of ions and predicting the chemical formula of the salts they form. Both highly electropositive and highly electronegative elements have a strong tendency to react. Moreover, the outer electrons will have less shielding from the nucleus, adding to the attractive force. The organization of elements on the periodic table allows for predictions concerning reactivity. Atoms with fewer electrons have a smaller, denser electron cloud with a stronger attraction towards the nucleus. Uranium-235 and uranium-238 occur naturally in the Earths crust. Carbon-12 is a stable isotope, while carbon-14 is a radioactive isotope (radioisotope). The number of electrons controls how tightly they are bound to the nucleus. Carbon 12 and Carbon 14 are both isotopes of carbon, one with 6 neutrons and one with 8 neutrons (both with 6 protons). You can effortlessly find every single detail about the elements from this single Interactive Periodic table.Polarizability Factors that Influence Polarizability 1. Let me tell you how this Interactive Periodic Table will help you in your studies.ġ). 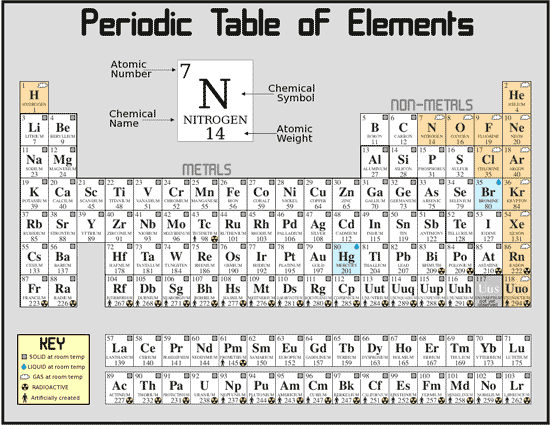 
Free Gift for you: Interactive Periodic Table In this way, the elements of the same group show similar chemical properties and they also have the same number of valence electrons. They are soft and can be cut easily with a kitchen knife.Īlso all the elements of group 1 have one valence electron.Īll the elements of group 18 are chemically inert (that means they do not easily react with other elements).Īnd all the elements of group 18 have a complete octet (that means they have 8 electrons in their outer shell). The elements lying in the same groups show similar chemical properties and they also have same number of valence electrons.Īll the elements of group 1 are highly reactive to water. There are total 18 vertical columns on periodic table. Groups are the vertical columns on the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
